The first year of Obamacare enrollment is officially in the books, and more than 7 million people enrolled for health insurance on a state or federally run Obamacare exchange. This figure more or less matches projected enrollment targets from the Obama administration in September. However, it seemed like a far cry away after fewer than 365,000 people had signed up during the first two months.

Source: White House on Flickr.
The companies that developed the architecture behind the federal and state Obamacare websites and the insurance companies have matured dramatically over these past six months. Consumers, as well, have grown more educated with regard to how Obamacare will affect them. The end result was a late surge in enrollment, surpassing everyone's expectations, and lending hope that 2015's enrollment which kicks off in mid-November will be a success, too.
There are, of course, still a number of unforeseen consequences that are left to play out from this first year of enrollment, such as whether or not insurance premiums will go up considerably next year and if we'll see more participation in 2015 from national insurers in the individual marketplaces.
Medical device makers draw the short end of the straw
One question, though, that a number of medical device companies were loudly answering for consumers and investors years ago was that the medical device excise tax, which is a 2.3% tax levied on medical device revenue would be an innovation killer.

Source: Steve Winton, Flickr.
I have to admit, I agreed wholeheartedly with the assumption that medical-device makers made prior to the full implementation of the Patient Protection and Affordable Care Act. The main thesis of medical device companies was that this tax would discourage innovation by increasing domestic costs, and could possibly push jobs overseas where these companies would be able to escape this tax.
Some CEOs and companies were quite proactive about the medical device excise tax, either cutting costs and jobs prior to its implementation or taking out op-eds in local publications to voice their opinions.
Medical device company Stryker, for example, announced in late 2011 that the additional costs from Obamacare, including the medical device excise tax, would cause it to cut 5% of its workforce, or nearly 1,200 jobs.
NuVasive CEO Alexis Lukianov went so far as to take out an op-ed column in the San Diego Union Tribune, predicting that he would need to cut planned new jobs and move portions of his manufacturing and R&D operations overseas to avoid the medical device excise tax.
Long story short, investors have been placing medical device companies in the loser's column since the word "Go."
Or did they...?
But that may not tell the entire story as an unintended consequence could actually wind up spurring innovation within the medical device industry as opposed to discouraging it. Keep in mind this is pure conjecture on my part (we'll need a year or two of data to determine if I'm right or wrong), but the enrollment data signifies to me that medical device companies could be in line to benefit from Obamacare.
At the heart of my assumption is the final surge in enrollment figures. I firmly believe that hospitals and outpatient facilities will directly benefit from more people being insured, leading to fewer instances where hospitals will be forced to write off revenue from uninsured and underinsured patients as uncollectable.
One thing to keep in mind is that being insured isn't a cure-all for hospitals and outpatient medical facilities. In order for the system to work we still need patients to pay their end of the out-of-pocket costs. While this clearly won't happen in every instance, I still expect a majority of consumers to honor paying their bills, thus reducing doubtful provisions for hospitals and boosting cash flow.
The thing with hospital cash flow is that it can be used for a myriad of activities, including building new medical care facilities, buying back stock, initiating or boosting a dividend, or purchasing new equipment for existing hospitals.
Given the uncertainty surrounding the implementation of Obamacare most hospitals have held back on making large equipment purchases. This extra cash flow generated by a lower rate of uninsured patients could be the perfect impetus for hospital operators to differentiate themselves from competitors with state-of-the-art equipment. In other words, while all medical device makers could ultimately benefit, those which make high-priced and specialized equipment could become the most direct beneficiaries.
Three medical device makers that may benefit
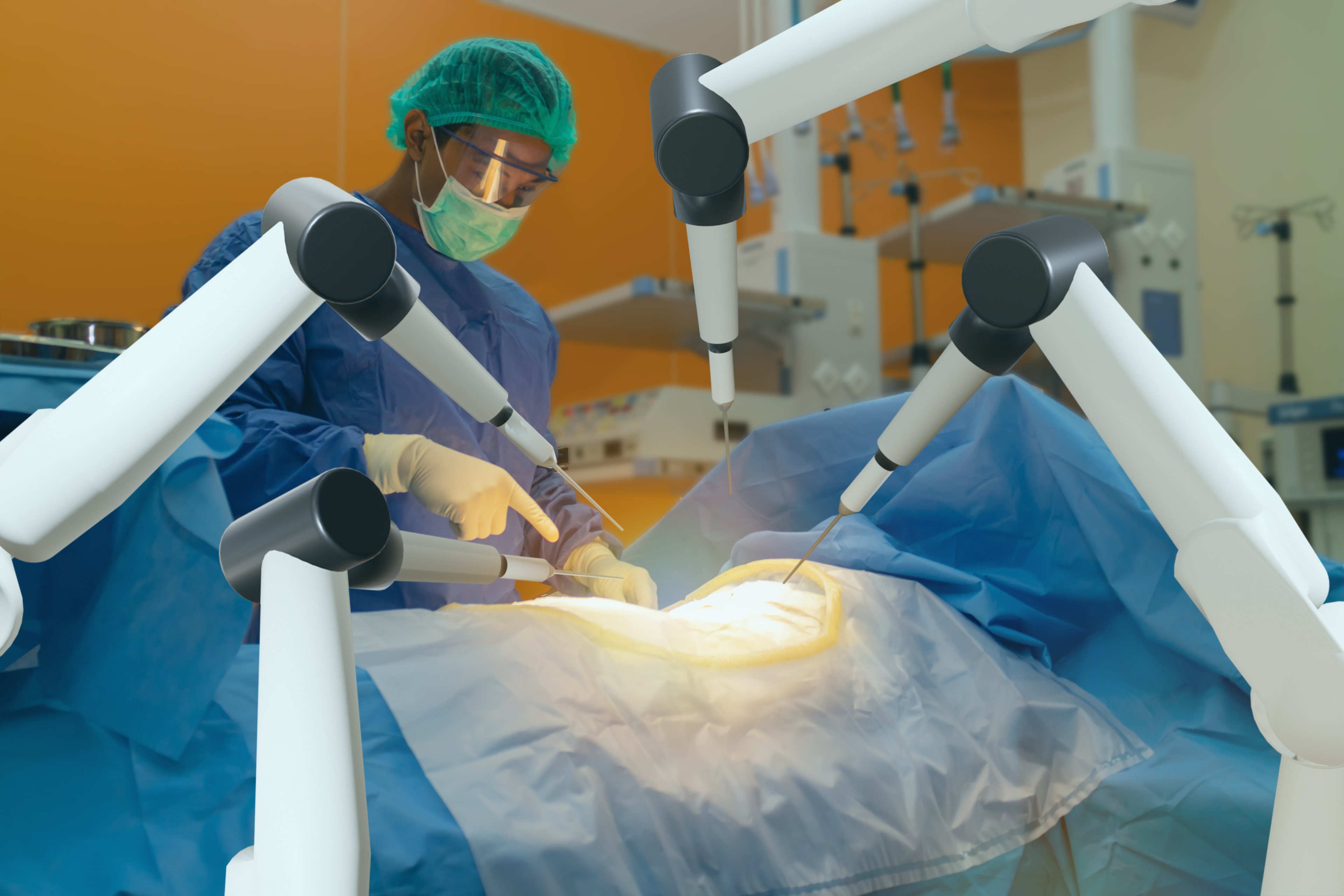
One potential beneficiary that stands out is robotic surgical system developer Intuitive Surgical (ISRG 2.35%), which is dealing with an ongoing investigation by the Food and Drug Administration into the safety of its da Vinci surgical systems in relation to standard laparoscopic surgery.

Source: Intuitive Surgical
Just two weeks ago Intuitive Surgical introduced its latest da Vinci system, known as the Xi, which comes with a wider range of motion, smaller and thinner arms, and better endoscopic architecture for clearer imaging. This system costs close to $2 million and a number of hospitals simply haven't been willing to shell out that type of cash for the highly efficient, minimally invasive soft tissue surgical device. Last week's enrollment surge could change that and really add some pep to the Xi's initial sales in the second quarter.
Another possible winner is Accuray (ARAY 8.10%), a small-cap developer of cancer-based radiosurgical devices. Array's two devices – CyberKnife, which detects tumors and corrects for patient movement during a radiation session for maximum dosing efficacy, and TomoTherapy, which accomplishes similar imaging goals – are not cheap. Various news sources have throughout the years listed the cost of the CyberKnife system as north of $4 million with its TomoTherapy system costing more than $3 million.

CyberKnife G4, Source: Accuray.
As you can see, it wouldn't take much to dramatically boost sales at Accuray if hospitals were willing to spend their cash. If there is indeed a doubtful provision drop as I suspect, the sheer need for more accurate cancer equipment given a rising number of diagnoses could present the perfect scenario for stronger sales at Accuray.
One final medical device maker worth watching, which is considerably more speculative than the previous two, is Hansen Medical (HNSN +0.00%). Hansen is a developer of the Sensei and Magellan robotic catheter system which allows physicians to manipulate and position catheters in patients. Similar to Intuitive Surgical, it profits from selling the physical machine itself as well as the services and products attached to its surgical system. At the moment Hansen is still in the early stages of its robotic system rollout, but the potential is there for rapid growth if hospitals suddenly have more disposable cash.
As I said, it'll take some time before we know how hospitals' cash flow responds to a lower uninsured rate, but I have a suspicion all the worry over medical device makers may have been for naught.