How would you feel about shelling out $750,000 for a year of treatment for a life-threatening disease?
Well, you're not alone.
Spinraza, the second drug to earn FDA approval from Ionis Pharmaceuticals (IONS 1.07%) is a potential life-saver, but the enormous list price set by its marketing partner, Biogen (BIIB 0.20%), received icy responses from some big American health insurers that will ultimately pay for it.

Image source: Getty Images.
Spinraza is the first available effective therapy for spinal muscular atrophy (SMA), a rare muscle-wasting disease. As SMA is the most common hereditary cause of infant mortality, end payers wouldn't dare refuse to cover its cost for all SMA patients, but many patients could find the pricey treatment out of reach. Two of the four largest publicly traded health insurers in the U.S., Anthem and Humana, have announced they will only cover the cost for patients with SMA type 1. Types 2 through 4 are less severe but comprise about half of the total SMA population.
Biogen badly needs a successful new drug launch. Could its big price tag be an unnecessary hurdle?
Initial restrictions
Although the FDA approved Spinraza for the treatment of SMA types 1 through 4, Anthem and Humana have announced decisions to limit coverage to those with the infantile-onset form, or type 1. America's largest insurer, UnitedHealth Group, is being a bit more generous by agreeing to cover types 1 through 3, with some restrictions.
America's third largest insurer, Aetna, hasn't weighed in yet, and Biogen investors will want to keep an eye open for its decision. With the drug's list price of $125,000 per dose, initiating treatment will run up a $750,000 bill in the first year, and then $375,000 each year thereafter for maintenance dosing.
Wanted: new blockbuster drugs
Biogen's product sales have been tapering off recently, and the company could really use a blockbuster drug launch to keep the needle moving in the right direction. As this would be one of the most expensive medicines on the planet, restrictions for just a handful of patients could significantly hinder Biogen's efforts to continue growing.
BIIB Revenue (TTM) data by YCharts
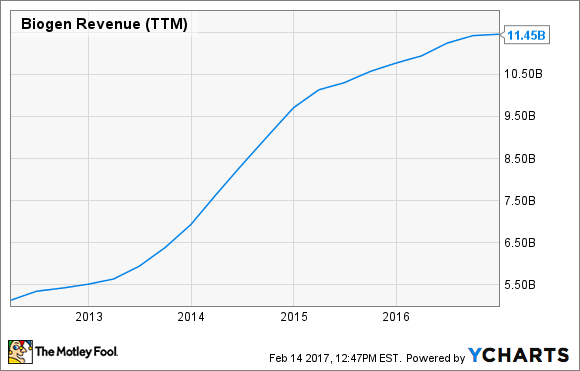
Ahead of its approval, peak annual sales estimates for Spinraza hovered between $1.0 billion and $1.5 billion. After the FDA granted its approval across the entire range of SMA patients, though, expectations that the drug's sales could top out around $2.0 billion and $2.5 billion each year have been the norm. Although Biogen reported $11.45 billion in total revenue last year, it looks as if insurer restrictions could become the difference between expansion or contraction of the company's top line in the years ahead.
Something's got to give
Type 1 SMA patients exhibit poor development of motor before they're 6 months old and never become strong enough to sit up on their own. Patients who fall under recent insurer restrictions are less severely affected, but their disease is far from trivial. Type 2 patients exhibit symptoms before they're 18 months old never gain the ability to stand on their own feet. Many with types 3 and 4 are eventually confined to wheelchairs and often develop comorbidities such as obesity and osteoporosis.
Spinraza looks like a public-relations disaster for both sides of the pricing debate, but I think restrictive insurers will bend before Biogen does. After all, public outrage could influence plan sponsors and individuals to switch insurance providers. Without any competing drugs, though, this really isn't an issue for the biotech.

Image source: Getty Images.
Perhaps the biggest factor informing Anthem's and Humana's decision to restrict coverage is a lack of available data. Biogen and Ionis ran placebo-controlled clinical trials with Spinraza in patients with types 1 and 2 only. It had reduced the risk of death or need for permanent ventilation by 47% compared with placebo at an interim analysis in the trial with type 1 patients. In the trial with type 2 patients, it significantly improved patients’ achievement of motor skill milestones.
Ionis and Biogen have additional data from patients with SMA types 1 through 3, but all the participants knew they were administering or receiving the actual drug. While patients with types 3 and 4 would probably benefit from the treatment, without a placebo-controlled study in these populations, we can't be entirely sure.
For now, insurers can point to the lack of available data in the restricted populations as an excuse not to provide coverage. Running additional trials with control arms is one way Biogen and Ionis could jump this hurdle, and plenty of positive real-world results from type 2 and 3 patients with UnitedHealth plans could be another. If you're holding shares of either biotech stock, you'll want to keep your eyes peeled for signs of both.