Image Source: Getty Images.
What happened
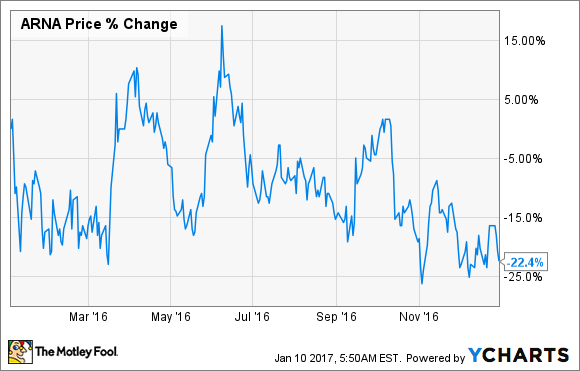
Like most biotechs, Arena Pharmaceuticals (ARNA +0.00%) had a rough 2016. The small-cap biotech's shares fell by a whopping 22.4% last year, according to data from S&P Global Market Intelligence.
Arena's stock suffered from both the poor commercial performance of its prescription obesity medication Belviq that's marketed by Japan's Eisai Co., as well as its heavy investment in developmental-stage products that resulted in the company losing around $20 million per quarter over the past 12 months.
So what
At the start of 2017, Arena decided to admit defeat on Belviq by transferring the remaining commercial rights to Eisai under an amended marketing and licensing agreement. Although the biotech will receive $23 million in cash and over $80 million in cost relief associated with Belviq's development per this modified agreement, this move essentially reverts Arena back into a clinical-stage company.
Now what
From a valuation standpoint, Arena's near-term prospects are now going to be mostly dependent on the fate of its mid-stage drug candidates etrasimod for ulcerative colitis and ralinepag for pulmonary arterial hypertension. Both drugs are expected to produce top-line data this year, which should determine if they progress into a pivotal-stage trial perhaps in early 2018.
While Arena is going after some big game from a commercial standpoint with its clinical pipeline -- etrasimod, for example, is targeting a market forecast to grow to an impressive $7 billion by 2021 -- there are ample reasons to remain cautious with this small-cap biotech this year. Even if both of these experimental drugs hit the mark in their ongoing mid-stage studies, pivotal-stage trials can easily take three to five years to complete.
Unfortunately, Arena's definitely going to need to raise capital well before then, based on its last-stated cash position of around $101 million, implying that more dilutive financing is nearly guaranteed in the years ahead. Even if Arena's shares do "pop" on a positive mid-stage result this year, this stock may have trouble sustaining any upward momentum due to the strong possibility of a sizable secondary offering coming into play.