Obesity is a big healthcare problem. In the U.S. alone, an estimated 35% of adults, or 85 million people, are believed to be obese, putting them at increased risk of certain forms of cancer, cardiovascular disease, and type 2 diabetes, among many other extremely serious medical conditions.

Source: Flickr via user Tony Alter.
For more than a decade, though, pharmaceutical options for overweight patients were limited primarily to over-the-counter products such as Orlistat because of serious safety issues that led the Food and Drug Administration to pull a number of drugs off the market. Since 2012, however, the FDA has now approved four new medications to aid in the fight against this growing pandemic.
With this in mind, let's take a deeper look at the two of the most prominent players in the obesity market today, Arena Pharmaceuticals (NASDAQ: ARNA) and Orexigen Therapeutics (NASDAQ: OREX), and consider the commercial potential of their respective FDA-approved diet drugs.

Source: Arena.
Arena Pharmaceuticals offers the appetite-suppressing drug Belviq
Belviq (lorcaserin) is a selective serotonin 5-HT2C agonist that regulates food intake by suppressing hunger, resulting in approximately 3%-4% of body weight loss, on average -- although about a quarter of patients in the clinical studies did see much higher levels of weight loss, on the order of 10%.
The drug was approved by the FDA in June 2012 and commercially launched by Eisai in the U.S. almost an entire year after its regulatory approval.
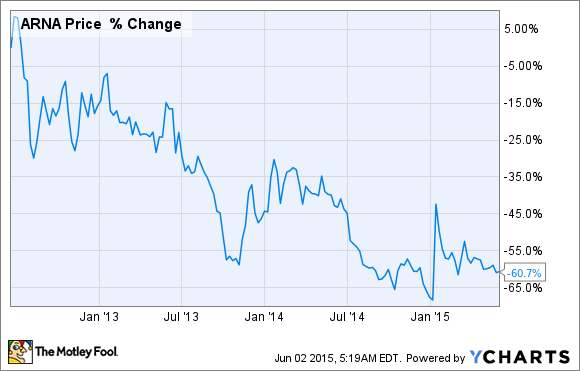
Because Belviq's efficacy for most patients is on the low side, the pill's commercial uptake has been painfully slow, crushing Arena's share price in the process:
Specifically, Arena and Eisai's first-quarter numbers suggest that Belviq will fall short of even $100 million in sales this year, despite being on the market for two years at this point. The two companies are hoping that new formulations, such as an extended-release version and/or a combo treatment with phentermine, will kick-start Belviq's U.S. launch, but only time will tell how this will play out.
Orexigen counters with Contrave
Contrave is a oral, sustained-release combination of bupropion and naltrexone designed to both suppress appetite and increase energy expenditure. The FDA approved the diet pill in Sept. 2014, and it was launched by Takeda Pharmaceuticals almost immediately afterwards. The drug was also recently approved in Europe under the brand name Mysimba, where Orexigen is actively seeking a marketing partner.

Source: Orexigen.
According to the FDA, the average patient taking Contrave lost 8% of his body weight in clinical trials, meaning it appears to be more effective as a weight-loss treatment than Belviq. The drug's safety profile is also somewhat better understood than Belviq, given that the company got about halfway through a large cardiovascular outcomes trial called the Light Study before the plug was pulled due to a data leak by Orexigen's management.
While the drug's commercial launch has so far been stronger than its competitors, Orexigen made a major misstep by publicly disclosing the interim data from the Light Study that directly led to the study's early demise. As a result, the FDA is now demanding another cardiovascular outcomes trial that could cost upwards of $200 million.
Takeda obviously wasn't thrilled at the prospect of shelling out hundreds of millions more for another post-marketing study, evinced by their decision to file a material breach of contract complaint against Orexigen. Orexigen therefore may end up without a marketing partner for Contrave -- and having to foot the bill for a massive clinical trial that it simply can't afford.
That's why the biotech's share price has been all over the map since Contrave's launch:
Key takeaways
Both of these fat-fighting pills are essentially indicated for the severely obese, or for overweight patients with one or more risk factors, such as diabetes or hypertension. The scope of the problem is such, though, that even this niche market should still encompass around 25 million adults in the U.S., and counting.
So, this latest generation of FDA-approved weight loss pills might seem like an amazing investing opportunity -- at least at first glance. A deeper look nonetheless shows that Belviq and Contrave may have a tough time reaching $1 billion in combined sales within the next decade. The truth is that neither pill is overwhelmingly effective, and the jury is still out on their long-term safety profiles.
All told, the size of the obesity market makes these two stocks worth watching going forward. But the fairly anemic launches of their respective diet pills bodes poorly for their long-term growth prospects, meaning something drastic would have to happen in order for them to transform into compelling growth stocks.