Short-sellers frequently targets small-cap biopharmas because their stocks can crumble in spectacular fashion when things don't go their way on the clinical or regulatory fronts. In fact, you'd be hard pressed to find more than a handful of small to mid-sized biopharmas that haven't attracted short-sellers in droves this year.

Source: Flickr via user Abd allah Foteih.
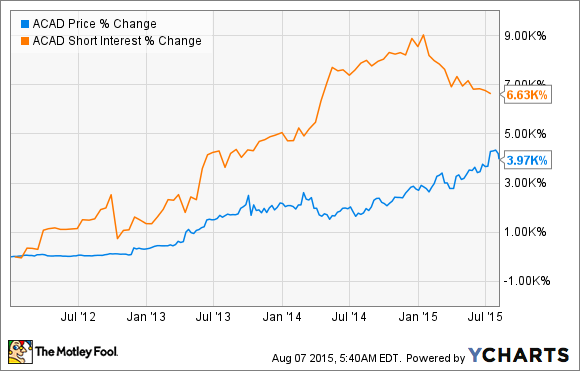
Nevertheless, these types of stocks can be a ticking time bomb for overenthusiastic shorts. Acadia Pharmaceutics (ACAD +0.14%), for example, saw a huge increase in short selling just ahead of its pivotal late-stage read out for its experimental Parkinson's disease psychosis drug Nuplazid. And as history has shown, shorts were terribly incorrect in their decision to dogpile on Acadia at this pivotal juncture. The odd part is they've continued to pour into this stock, all while its share price has pushed higher and higher -- causing shorts even more pain in the process:
Despite this somewhat brutal example, shorts have still poured into some of Acadia's more promising contemporaries such as Bellicum Pharmaceuticals (BLCM +0.00%), Exelixis (EXEL 1.92%), and Sarepta Therapeutics (SRPT +3.29%). Even if I could ignore the questionable morality of shorting stocks working on life-saving diseases, I still wouldn't dream of shorting these three stocks. Here's why.
Bellicum might leapfrog its competitors in the CAR-T space
Bellicum is lagging behind its peers, including Juno Therapeutics and Kite Pharma, in the CAR-T space for blood cancers and solid tumors. But its novel approach toward developing the next generation of CAR-T products could enable it to one day dominate this multibillion-dollar market.
In a nutshell, CAR-Ts have produced some promising early stage results in a host of blood cancers, but they've also been linked to several serious -- and even deadly -- adverse events. Bellicum is hoping to lower the potential for serious adverse events through its CaspaCIDe safety switch inserted in its CAR-T products. This molecular switch can be triggered by infusing a small-molecule called rimiducid into patients exhibiting signs of toxicity, thereby causing the cells to undergo programmed cell death.
In another stroke of genius, Bellicum developed a second molecule switch designed to "amp up" the therapeutic response in the presence of tumor cells, giving the company a relatively unique ability to modulate the activity of its CAR-T constructs in vivo.
Short-sellers appear to be unimpressed by Bellicum's platform, however, gobbling up 13% of the float at last count. With the company set to begin human trials in the first half of 2016, I wouldn't be surprised if they begin moving to the sidelines. After all, there has been a ton of high-dollar licensing activity in this space of late, and Bellicum is probably going to follow suit in this regard.
Exelixis is soaring after its flagship drug breaks through in kidney cancer
Exelixis got a much-needed boost last month, after its main value driver, Cometriq, reported strong late-stage results as a second-line treatment in kidney cancer. Specifically, Cometriq met its primary endpoint of improving progression-free survival for patients with renal-cell carcinoma, or kidney cancer, compared with those receiving Novartis' Afinitor (everolimus). Even though this positive trial result should put Cometriq on track toward taking a big slice out of Afinitor's $400 million-plus in sales moving forward, shorts haven't exactly backed off, with a nearly a quarter of the float being short, per the latest figures.

Source: Exelixis.
With Exelixis and its partner Roche widely expected to receive good news from the FDA in November for their experimental metastatic melanoma combo therapy of cobimetinib and Zelboraf, and the drugmaker shoring up its cash position with a recent $135 million offering, I think the future looks particularly bright for this small-cap biopharma. Overall, Exelixis is shaping up into an attractive takeover target after these key clinical and business developments, putting shorts into a rather precarious, high-risk position moving forward.
Sarepta's stock could soar with a regulatory approval for its experimental DMD drug
Sarepta's shares have already climbed by a staggering 139% this year, after the company announced that the FDA agreed to review its exon-skipping Duchenne muscular dystrophy, or DMD, drug, eteplirsen. Shorts, though, still hold a massive position in the stock, with over 35% of the float being sold short. As a reminder, eteplirsen's regulatory pathway has been hindered by the fact that Sarepta has only completed a midstage study that included a grand total of 12 patients.
While there's a good chance that the FDA could still decide to reject the application outright or issue a Complete Response Letter following a formal review, the obvious risk for short-sellers is that eteplirsen's limited data turn out to be enough to convince the agency that its benefits outweigh the risks for a disease that is always fatal. Perhaps the biggest issue that should be keeping short-sellers up at night is that an eteplirsen approval may validate Sarepta's remaining DMD pipeline that seeks to treat upwards of 80% of this patient population -- meaning that its DMD platform has the potential to rake in billions in sales.
Are any of these three stocks compelling buys now?
Although I wouldn't be caught shorting any of these small-cap biopharmas because of their explosive upside potential, Exelixis is perhaps the only clear-cut buyout of the group. Exelixis already has an approved product on the market in Cometriq, and it's finally starting to blossom into a viable oncology company with the drug's positive top-line results in kidney cancer. Bellicum and Sarepta, by contrast, are probably best viewed as intriguing watchlist candidates because of the speculative nature of their upcoming catalysts.