No matter what your reasons for investing may be, you would probably be thrilled with a stock that turns a relatively small amount of money -- say, $50,000 -- into a million dollars. Finding companies of this caliber can be a daunting task: There are near-endless options on the market, but most stocks will never deliver these kinds of gains.
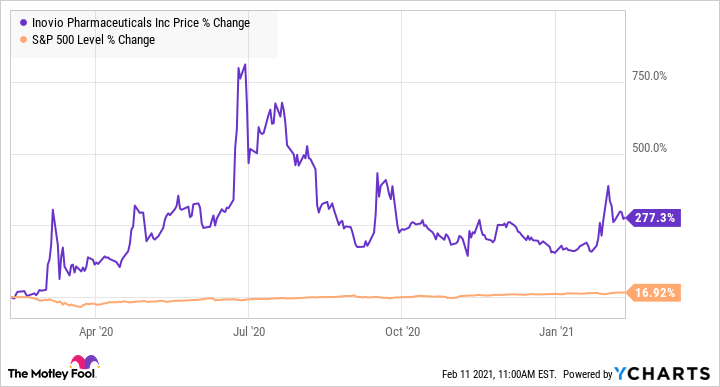
Today, let's take a closer look at clinical-stage biotech company Inovio Pharmaceuticals (INO -37.85%) and see whether it has what it takes to join that select group of millionaire-maker stocks. Inovio rose to prominence last year thanks to its efforts to develop a vaccine for COVID-19, and its shares are up by 277% over the past 12 months as a result, compared to gains of 16% for the S&P 500 in the same period. Can the biotech keep up the momentum moving forward?
Where Inovio's coronavirus-related efforts stand
Inovio's experimental COVID-19 vaccine, INO-4800, is a DNA vaccine. It contains a plasmid (circular DNA molecules within a pathogen) that carries parts of the genetic code of the SARS-CoV-2 virus that causes COVID-19. INO-4800 is administered intradermally using Inovio's proprietary device called Cellectra 2000. Once inside a patient's skin, it can trigger an immune response that confers protection against the SARS-CoV-2 virus. In a phase 1 clinical trial, INO-4800 generated neutralizing antibodies or T-cell responses in 100% of the participants.Antibodies are proteins that defend healthy cells from pathogens. T-cells are white blood cells that bind to and instruct virus-infected cells to self-destruct.
Inovio is currently running a phase 2/3 clinical trial for its candidate. The phase 2 portion of the study started in early December. It will evaluate the safety, tolerability, and immunogenicity (the ability to provoke an immune response in the body) of the vaccine in about 400 participants in the U.S.
Note that the phase 3 segment of this study remains on partial clinical hold due to the U.S. Food and Drug Administration (FDA) having concerns regarding the Cellectra 2000 device.

Image source: Getty Images.
Meanwhile, the company started a separate phase 2 study in China in collaboration with Advaccine Biopharmaceuticals Suzhou. This study will enroll about 640 patients and will test the safety and immunogenicity of INO-4800.
Inovio could still go on to make a dent in this market if its vaccine proves more effective than alternatives or if it has some other advantages over its competitors. The only such advantage (for now) is that it can be stored at room temperature for up to a year.
Both vaccines that have been granted emergency use authorization (EUA) by the FDA need to be kept at extremely cold temperatures, which has resulted in logistical difficulties. But Inovio is still some distance away from even showing that its vaccine is effective. It is hard to justify purchasing shares of the company today based solely on its coronavirus-related efforts.
What else does Inovio Pharmaceuticals have to offer?
Inovio boasts a little more than a dozen products in its pipeline. Arguably the most promising of the bunch (other than INO-4800) is VGX-3100, a potential treatment for a human papillomavirus (HPV)-associated precancerous condition called cervical dysplasia. VGX-3100 is currently in a phase 3 clinical trial.
The company had planned to release data from this trial late last year, but the pandemic disrupted the study. Inovio now expects to provide some data from this study in the first half of this year.
In a phase 2 clinical trial, VGX-3100 led to a 25% or more reduction in HPV-associated high-grade squamous intraepithelial lesion (an area of abnormal cells that form on certain organs as a result of HPV) in 63% of the participants at six months after treatment.
According to Inovio, the annual incidence of cervical dysplasia is 195,000 people in the United States and 233,000 people in Europe. There are currently no FDA-approved medicines for it, and if VGX-3100 ends up receiving regulatory clearance, it could turn into a cash cow for Inovio.
Is Inovio going to make you a millionaire?
In my view, Inovio doesn't look likely to be a millionaire-maker stock from here on out. Too much has to go right in order for that to happen: Its leading programs, INO-4800 and VGX-3100, would both have to go on to receive FDA approval and capture a large share of their respective markets. But since the COVID-19 vaccine segment is highly competitive, it isn't clear to what extent INO-4800 will make a dent in the market, even if it is eventually approved by the FDA.
VGX-3100 is promising, but it can still run into regulatory roadblocks or fail to prove effective in its ongoing phase 3 study. Given these factors, Inovio seems slightly on the risky side, and I think most investors looking to be made into millionaires should consider other options. There is a slew of alternatives for those looking for exposure to coronavirus vaccine stocks or equally exciting biotech picks.