The coronavirus vaccine developed by AstraZeneca (AZN +0.00%) in partnership with Oxford University has succeeded in preventing transmission of COVID-19. Unfortunately, a disappointing series of missteps by the company and its collaboration partner have irreparably damaged the vaccine's image in the eyes of the public.
Before writing AstraZeneca off completely, investors would do well to remember that this is a drug company with a red-hot oncology segment. Add up just a handful of the growth drivers in AstraZeneca's product lineup, and the company's missteps with its coronavirus vaccine seem like a minor hiccup.

Image source: Getty Images.
Reasons to buy AstraZeneca now
AstraZeneca managed to grow its top and bottom lines by double-digit percentages in 2020, thanks to label-expanding approvals for cancer therapies that will continue driving growth in 2021. Tagrisso is a popular tablet that earned approval in December to treat lung cancer patients who caught the disease in its early stage and had their tumors surgically removed.
Label expansions to prevent the recurrence of cancer are highly coveted by drugmakers, because these patients tend to stay in treatment a long time. Tagrisso sales surged 36% to $4.3 billion in 2020, and new long-term patients will allow this revenue stream to grow at a blazing pace for at least a few more years.
Tagrisso is arguably AstraZeneca's most important drug right now, but it isn't the only blockbuster cancer therapy in the company's lineup with a bright future. Lynparza is a tablet that first earned approval in 2014 to treat relapsed ovarian cancer patients; it has since expanded its addressable patient population to include patients with breast, prostate, and pancreatic cancers. Lynparza sales jumped 24% year over year to $2.2 billion in 2020, and the drug's on pace to generate more than $4 billion in 2021.
Enhertu is a more recently launched cancer drug with enormous potential for people with disease driven by HER2 mutations; it was first approved by the Food and Drug Administration in 2019, for breast cancer patients who relapse after treatment with drugs like Herceptin. Sales of this future blockbuster are just beginning to gain steam. Earlier this year, the FDA approved Enhertu to treat stomach cancer patients in the third-line setting, after the drug reduced their risk of death by 41% compared to standard chemotherapy.
Calquence is an increasingly popular tablet for leukemia that AstraZeneca launched in 2018, and it's gaining on Imbruvica, an older treatment from Johnson & Johnson (JNJ 0.10%) and AbbVie (ABBV 1.79%) that works the same way. Sales of Imbruvica finished 2020 on pace to exceed a combined $10 billion annually for AbbVie and Johnson & Johnson, but investors will see the market for this drug class shift toward Calquence. Clinical trial results that AstraZeneca released earlier this year showed a significant safety advantage for Calquence in a head-to-head showdown with Imbruvica.
Look further ahead
The COVID-19 vaccine from AstraZeneca and Oxford University hasn't been authorized in the U.S. yet, but the phase 3 data available right now looks pretty good any way you slice it. Sadly, a long string of unnecessary blunders destroyed the vaccine's reputation in the eyes of the public.
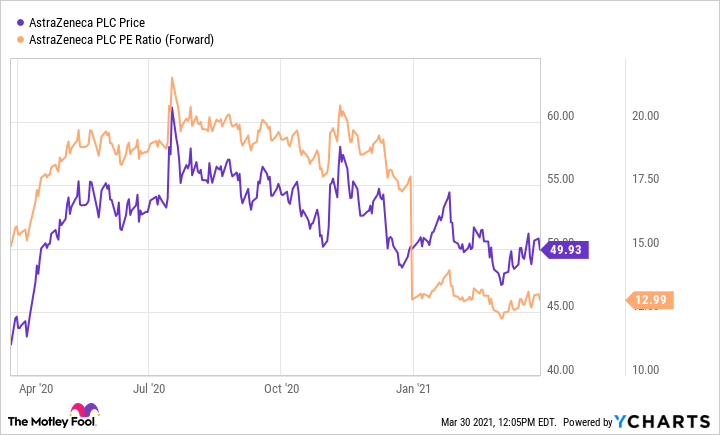
AstraZeneca took on the challenge of developing a COVID-19 vaccine with the intention of selling it at break-even prices. This didn't stop the stock from rising last summer when it looked like the company was leading the race:
Most of those gains have since been wiped out, despite soaring cancer-drug sales that have pushed up the company's earnings outlook. At recent prices, AstraZeneca has been trading at the ultralow price of 13 times forward earnings expectations.
AstraZeneca's vaccine blunders are pressuring the stock right now, but it's just a matter of time before the company's stellar performance in the oncology field gets reflected in the share price. If you're willing to look past bad vaccine news that will soon be forgotten, buying this stock now and holding it for the long run looks like a great way to build wealth.