Investing in the stock market has historically been one of the most important pathways to financial success. As you dive into researching stocks, you'll often hear them discussed with reference to different categories of stocks and different classifications. Here are the major types of stocks you should know.
List | Type |
|---|---|
1 | Common stock |
2 | Preferred stock |
3 | Large-cap stocks |
4 | Mid-cap stocks |
5 | Small-cap stocks |
6 | Domestic stock |
7 | International stocks |
8 | Growth stocks |
9 | Value stocks |
10 | IPO stocks |
11 | Dividend stocks |
12 | Non-dividend stocks |
13 | Income stocks |
14 | Cyclical stocks stocks |
15 | Non-cyclical stocks |
16 | Safe stocks |
17 | ESG stocks |
18 | Blue chip stocks |
19 | Penny stocks |
Common stock and preferred stock
Most stock that people invest in is common stock. Common stock represents partial ownership in a company, with shareholders getting the right to receive a proportional share of the value of any remaining assets if the company is dissolved. Common stock gives shareholders theoretically unlimited upside potential, but they also risk losing everything if the company fails without having any assets left over.
Preferred stock works differently by giving shareholders a preference over common shareholders to get back a certain amount of money if the company dissolves. Preferred shareholders also have the right to receive dividend payments before common shareholders. The net result is that preferred stock as an investment often more closely resembles fixed-income bond investments than regular common stock. Often, a company will offer only common stock. This makes sense, since it's most often what shareholders want to buy.
Large-cap, mid-cap, and small-cap stocks
Stocks also get categorized by the total worth of all their shares, which is called market capitalization. Companies with the biggest market capitalizations are called large-cap stocks, with mid-cap and small-cap stocks representing successively smaller companies.
There's no precise line that separates these categories from each other. However, one often-used rule is that stocks with market capitalizations of $10 billion or more are treated as large-caps, with stocks having market caps between $2 billion and $10 billion qualifying as mid-caps, and stocks with market caps less than $2 billion treated as small-cap stocks.
Large-cap stocks are generally considered safer and more conservative as investments, while mid-cap and small-cap stocks have a greater capacity for future growth but are riskier. However, just because two companies fall into the same category here doesn't mean they have anything else in common as investments or that they'll perform in similar ways in the future.
Make your portfolio reflect your best vision for our future.
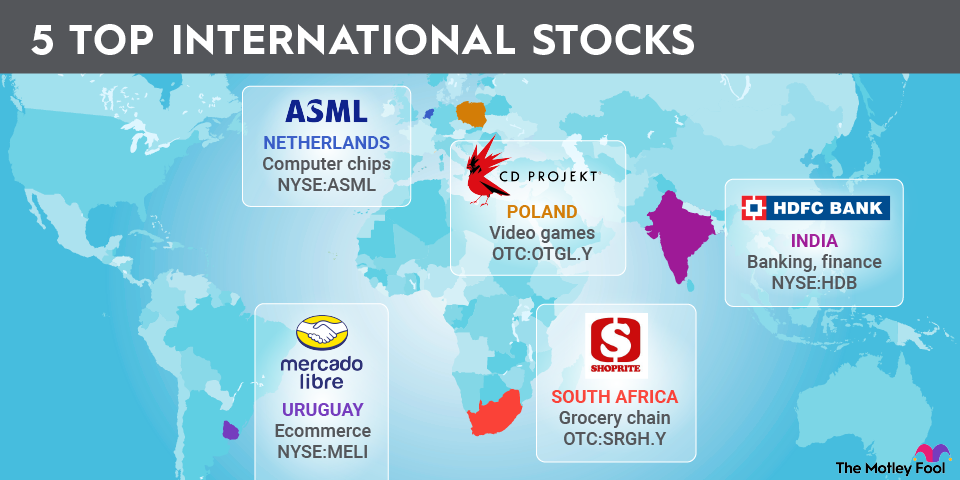
Domestic stocks and international stocks
You can categorize stocks by where they're located. For purposes of distinguishing domestic U.S. stocks from international stocks, most investors look at the location of the company's official headquarters.
However, it's important to understand that a stock's geographical category doesn't necessarily correspond to where the company gets its sales. Philip Morris International (PM -0.35%) is a great example. Its headquarters are in the U.S., but it sells its tobacco and other products exclusively outside the country. Especially among large multinational corporations, it can be hard to tell from business operations and financial metrics whether a company is truly domestic or international.
Growth stocks and value stocks
Another categorization method distinguishes between two popular investment methods. Growth investors tend to look for companies that are seeing their sales and profits rise quickly. Value investors look for companies whose shares are inexpensive, whether relative to their peers or to their own past stock price.
Growth stocks tend to have higher risk levels, but the potential returns can be extremely attractive. Successful growth stocks have businesses that tap into strong and rising demand among customers, especially in connection with longer-term trends throughout society that support the use of their products and services. Competition can be fierce, though, and if rivals disrupt a growth stock's business, it can fall from favor quickly. Sometimes, even just a growth slowdown is enough to send prices sharply lower, as investors fear that long-term growth potential is waning.
Value stocks, on the other hand, are seen as being more conservative investments. They're often mature, well-known companies that have already grown into industry leaders and don't have as much room left to expand further. Yet, with reliable business models that have stood the test of time, they can be good choices for those seeking more price stability while still getting some of the positives of exposure to stocks.
IPO stocks
IPO stocks are stocks of companies that have recently gone public through an initial public offering (IPO). They often generate a lot of excitement among investors looking to get in on the ground floor of a promising business concept. But they can also be volatile, especially when there's disagreement within the investment community about their prospects for growth and profit. A stock generally retains its status as an IPO stock for at least a year and for as long as two to four years after it becomes public.
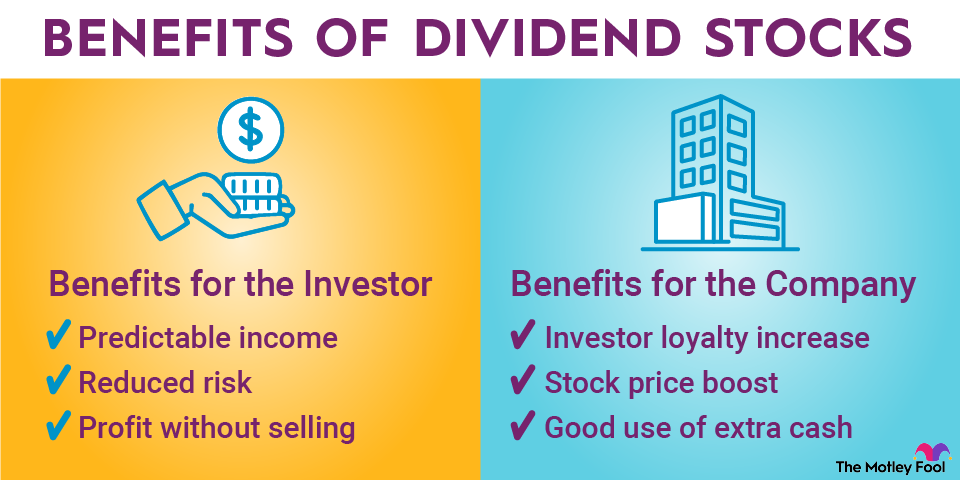
Dividend stocks and non-dividend stocks
Many stocks make dividend payments to their shareholders on a regular basis. Dividends provide valuable income for investors, which is why dividend stocks are highly sought after among certain investment circles. Technically, paying even $0.01 per share qualifies a company as a dividend stock.
However, stocks don't have to pay dividends. Non-dividend stocks can still be strong investments if their prices rise over time. Some of the biggest companies in the world don't pay dividends, although the trend in recent years has been toward more stocks making dividend payouts to their shareholders.
Income stocks
Income stocks are another name for dividend stocks, as the income that most stocks pay out comes in the form of dividends. However, income stocks also refer to shares of companies that have more mature business models and have relatively fewer long-term opportunities for growth. Ideal for conservative investors who need to draw cash from their investment portfolios right now, income stocks are a favorite among those in or nearing retirement.
Cyclical stocks and non-cyclical stocks
National economies tend to follow cycles of expansion and contraction, with periods of prosperity and recession. Certain businesses have greater exposure to broad business cycles, so investors refer to them as cyclical stocks.
Cyclical stocks include shares of companies in industries like manufacturing, travel, and luxury goods, because an economic downturn can take away customers' ability to make major purchases quickly. When economies are strong, however, a rush of demand can make these companies rebound sharply.
By contrast, non-cyclical stocks, also known as secular or defensive stocks, don't have those big swings in demand. An example of non-cyclical stocks would be grocery store chains because no matter how good or bad the economy is, people still have to eat. Non-cyclical stocks tend to perform better during market downturns, while cyclical stocks often outperform during strong bull markets.
Safe stocks
Safe stocks are stocks whose share prices make relatively small movements up and down compared with the overall stock market. Also known as low-volatility stocks, safe stocks typically operate in industries that aren't as sensitive to changing economic conditions. They often pay dividends as well, and that income can offset falling share prices during tough times.
Stocks categorized by sector
You'll often see stocks broken down by the type of business they're in. The basic categories most often used include stock market sectors:
- Communication Services -- Telephone, internet, media, and entertainment companies
- Consumer Discretionary -- Retailers, automakers, and hotel and restaurant companies
- Consumer Staples -- Food, beverage, tobacco, and household and personal products companies
- Energy -- Oil and gas exploration and production companies, pipeline providers, and gas station operators
- Financial -- Banks, mortgage finance specialists, and insurance and brokerage companies
- Healthcare -- Health insurers, drug and biotech companies, and medical device makers
- Industrial -- Airline, aerospace and defense, construction, logistics, machinery, and railroad companies
- Materials -- Mining, forest products, construction materials, packaging, and chemical companies
- Real Estate -- Real estate investment trusts (REITs), and real estate management and development companies
- Technology -- Hardware, software, semiconductors, communications equipment, and IT services companies
- Utilities -- Electric, natural gas, water, renewable energy, and multi-product utility companies
ESG stocks
ESG investing refers to an investment philosophy that puts emphasis on environmental, social, and governance concerns. Rather than focusing entirely on whether a company generates profit and is growing its revenue over time, ESG principles consider other collateral effects on the environment, company employees, customers, and shareholder rights.
Tied to ESG's governing rules is socially responsible investing, or SRI. Investors using SRI screen out stocks of companies that don't match up to their most important values. However, ESG investing has a more positive element; rather than just excluding companies that fail key tests, it actively encourages investment in specific companies. With evidence showing that a clear commitment to ESG principles can improve investing returns, there's a lot of interest in the area.
Blue chip stocks
Finally, there are stock categories that make judgments based on perceived quality. Blue chip stocks tend to be the cream of the crop in the business world, featuring companies that lead their respective industries and have gained strong reputations. They typically don't provide the absolute highest returns, but their stability makes them favorites among investors with a lower tolerance for risk.
Types of stock classes
Companies may issue different classes of stock with varying voting rights and access to dividends. The main purpose of creating different stock classes is to separate economic ownership from control and to tailor voting rights and other privileges to different investor groups.
This allows companies to raise capital from public markets without necessarily giving up influence to outside investors. For instance, companies may create Class A shares with more voting rights than other classes (like Class B or Class C), which can allow founders, executives, or specific investors to retain dominance even after the company becomes publicly traded.
Choosing stocks for your portfolio
Choosing stocks for your investment portfolio is a critical step towards achieving your financial goals, whether it's long-term growth, wealth preservation, or generating income. However, it's important to carefully assess your investment interests, risk tolerance level, and portfolio objectives before you put cash to work in stocks.
You should have a keen understanding of your long-term investment goals. Focus on quality companies with strong leadership teams and durable growth tailwinds based on a set of products and/or services that face prolonged demand.
Look for businesses with consistent revenue and earnings growth, or at least a defined path to profitability if a business you're evaluating isn't yet profitable. It's often best to diversify your cash into a wide range of stocks and sectors so that you can benefit from the growth profiles of different businesses in a variety of market environments.
Whether that's investing in a combination of growth stocks, value stocks, dividend stocks, blue chip stocks, or otherwise, make sure you understand the business you are buying and ensure it fits with the preferred balance and construction of your portfolio. A long-term growth horizon of at least three to five years for any stocks you buy is key.
Penny stocks
Penny stocks are low-quality companies whose stock prices are extremely inexpensive, typically less than $1 per share. With dangerously speculative business models, penny stocks are prone to schemes that can drain your entire investment. It's important to know about the dangers of penny stocks.
You've probably heard that portfolio diversification is important for developing strong, stable investments. Keep all of these stock classifications in mind as you plan for diversity -- investing across companies of different market capitalizations, geographies, and investing styles contributes to a well-balanced portfolio.
How to research different types of stocks
You can research different types of stocks through numerous aspects of fundamental analysis. Fundamental analysis is a method to determine a stock's intrinsic value by examining factors that affect a company's business.
For example, reviewing a company's financial statements, such as the balance sheet, income statement, and statement of cash flow, is a key part of fundamental analysis. Key metrics, including revenue, earnings per share (EPS), the price-to-earnings (P/E) ratio, and the debt-to-equity ratio, should factor into your fundamental analysis, too.
You’ll also want to evaluate qualitative factors like the management team, brand recognition, industry trends, and the company's competitive advantage. Different types of stocks will require you to focus on additional specific characteristics when you do your research.
For example, growth stocks that are expected to outperform the market are best researched by looking for companies with growing revenues and earnings. Similarly, defensive stocks offer consistent returns regardless of economic conditions, so research into these businesses should focus on stable companies in non-cyclical industries like consumer staples.
Related investing topics
Future trends: Which types of stocks might dominate tomorrow?
Stocks in sectors driven by innovation and addressing global challenges are poised to dominate the future. Key areas for potential long-term growth include artificial intelligence (AI) and technology broadly, healthcare, e-commerce, and financial technology (fintech), just to name a handful.
AI is a transformative force that's enabling smarter decisions and automating tasks across almost every industry, from finance to manufacturing. Companies providing AI infrastructure (like semiconductors), software, and services are expected to see sustained, high growth.
As for healthcare, growth in this space is being driven by factors ranging from an aging population to technological advancements. The healthcare sector is increasingly focused on personalized medicine, advanced diagnostics, and new therapies.
The shift to online retail and digital services continues to accelerate, driven by consumer demand for convenience and personalized experiences. Logistics and cybersecurity companies that support this digital infrastructure are also in high demand.
Then there's innovation in digital payments, decentralized finance (DeFi), and AI-driven financial services that's fundamentally changing how people manage money. This is also reshaping the fintech sector and its long-term potential.