Everybody needs healthcare -- or will, at some point. And, when there’s something everyone needs, there’s a huge opportunity for investors. Healthcare stocks are shares of publicly traded companies that provide healthcare products or services.
About $9.8 trillion is spent on healthcare globally. Half of that amount -- around $4.9 trillion -- is spent in the U.S. With the healthcare sector growing significantly faster than the overall economy, the numbers will almost certainly be much larger by the end of the decade.
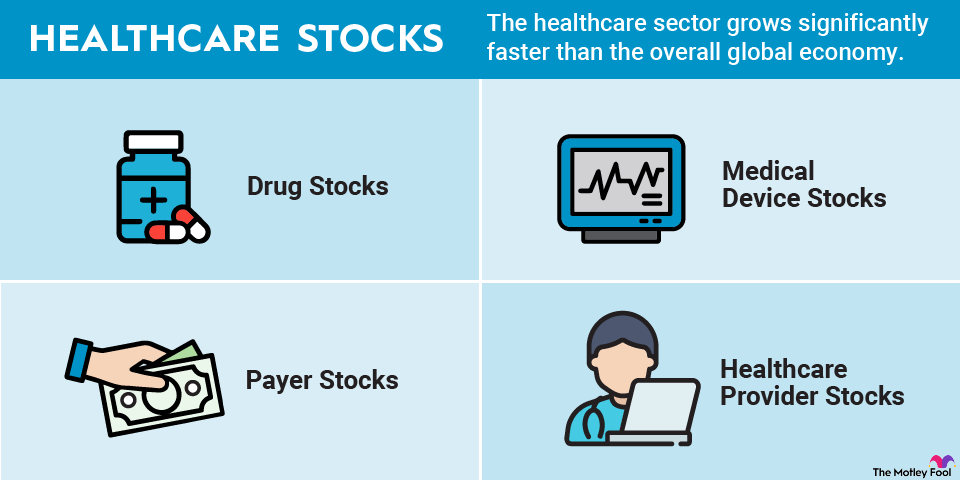
How can investors profit from this growth? Here’s what you need to know about investing in healthcare stocks.
Different types of healthcare stocks
The healthcare sector is so broad that there are several different kinds of healthcare stocks. Four of the most important types are:
- Drug stocks: Drugmakers focus on developing drugs that treat or prevent diseases. Biotech companies use live organisms such as bacteria or enzymes to develop drugs, while pharmaceutical companies use chemicals. Drug stocks range from huge companies with billions of dollars in sales each year to small biotech firms with no products on the market yet.
- Medical device stocks: Medical device companies make devices used to care for patients. The devices range from disposable gloves and thermometers to artificial heart valves and robotic surgical systems. Medical device stocks include many health tech stocks, as well as medical equipment stocks.
- Payer stocks: Payers, including health insurers and pharmacy benefit managers (PBMs), play an especially important role in the U.S. healthcare system. Insurers charge premiums to individuals and employers to pay for healthcare costs, while PBMs administer prescription drug benefits for employers and health plans.
- Healthcare provider stocks: Healthcare providers stand at the front lines, delivering healthcare services to patients. They include hospitals, physician practices, home health companies, and long-term care facilities.
Top healthcare stocks to buy in 2026
Strong companies can be found within each type of healthcare stock. Here are some top healthcare stocks to buy that represent each type:
| Name and ticker | Market cap | Dividend yield | Industry |
|---|---|---|---|
| Vertex Pharmaceuticals (NASDAQ:VRTX) | $117.7 billion | 0.00% | Biotechnology |
| Intuitive Surgical (NASDAQ:ISRG) | $207.8 billion | 0.00% | Healthcare Equipment and Supplies |
| TransMedics Group (NASDAQ:TMDX) | $4.7 billion | 0.00% | Healthcare Equipment and Supplies |
| UnitedHealth Group (NYSE:UNH) | $311.6 billion | 2.54% | Healthcare Providers and Services |
| HCA Healthcare (NYSE:HCA) | $107.7 billion | 0.61% | Healthcare Providers and Services |
| Cardinal Health (NYSE:CAH) | $47.8 billion | 1.01% | Healthcare Providers and Services |
1. Vertex Pharmaceuticals

NASDAQ: VRTX
Key Data Points
Vertex Pharmaceuticals (VRTX -1.24%) stands out as one of the top biotech stocks on the market. The company primarily focuses on developing drugs that treat the underlying cause of cystic fibrosis (CF), a rare genetic disease that damages the lungs and other organs. Vertex has also expanded beyond CF with gene-editing therapy Casgevy (a one-time treatment for the rare blood disorders sickle cell disease and transfusion-dependent beta-thalassemia) and acute pain therapy Journavx.
2. Intuitive Surgical

NASDAQ: ISRG
Key Data Points
Intuitive Surgical (ISRG +0.20%) is a great example of a medical device stock that also falls into the category of surgical stocks. The company’s Da Vinci robotic surgical system has been used in more than 17.6 million procedures since its 1999 introduction. Intuitive Surgical also markets Ion, a robotic system for minimally invasive peripheral lung biopsy. Over the long run, the company should have tremendous growth opportunities ahead with an aging population requiring the types of surgical procedures for which Da Vinci is frequently used.
3. TransMedics Group

NASDAQ: TMDX
Key Data Points
TransMedics Group (TMDX -0.20%) developed and sells the Organ Care System (OCS), a system for transporting lungs, hearts, and livers intended for transplantation. OCS replicates the human body in many ways, keeping donated organs alive until they can be transplanted. TransMedics also created a National OCS Program (NOP) that provides end-to-end services related to organ transplantation, including air transportation. The company has a major growth opportunity in disrupting the organ transplant market.
4. UnitedHealth Group

NYSE: UNH
Key Data Points
UnitedHealth Group (UNH -0.83%) ranks as the largest commercial health insurer in the U.S. It also operates one of the biggest PBMs and is a leader in healthcare delivery services. The company’s size, stability, and dividend make UnitedHealth Group one of the most attractive payer stocks on the market. UnitedHealth Group has also expanded its presence in the healthcare provider market with its 2023 acquisition of home health services provider LHC Group.
5. HCA Healthcare

NYSE: HCA
Key Data Points
HCA Healthcare (HCA -1.45%) owns and operates 190 hospitals and around 2,500 outpatient facilities, including surgery centers, physician clinics, freestanding emergency rooms, and urgent care centers. It's the largest health system operator in the U.S. HCA also operates healthcare facilities in the United Kingdom. The company continues to grow, in large part, by making acquisitions.
6. Cardinal Health

NYSE: CAH
Key Data Points
Cardinal Health (CAH -0.68%) distributes branded and generic pharmaceuticals, medical and laboratory products, and supply chain services to healthcare providers. The company serves over 90% of U.S. hospitals and operates in more than 30 countries.
What to look for in healthcare stocks
How do you find the absolute best healthcare stocks to buy? There are four key things to look for:
1. Growth prospects
- Determine how quickly revenue has grown in recent years.
- Read the investor presentations on companies’ websites to learn their strategies for growth and the size of their potential markets.
- Check out the companies’ rivals to see if their strategies seem to be as good or better.
- Don’t overlook the possibility that mergers and acquisitions (M&A) could boost a company’s growth prospects.
2. Financial strength
- Ideally, a company will already be profitable. If it isn’t, make sure you learn how it plans to achieve profitability and how quickly it expects to do so.
- Look at the company's cash position on its balance sheet (a financial statement that lists all the company’s assets, liabilities, and shareholder equity) in the company's annual and quarterly regulatory filings. The larger the cash position, the better.
- Evaluate the free cash flow (FCF) generated by a company. FCF is the cash left over after operating expenses and capital expenditures (which include money spent on buildings, equipment, and land). As with the cash position, the higher a company’s FCF, the stronger its financial position.
Your dollars and mine, our capital, is helping shape the world.
3. Valuation
- Determine the value of a healthcare stock before buying to make sure you're paying a fair price.
- The price-to-earnings (P/E) ratio is the most popular valuation metric. It measures the price of a stock in relation to its earnings per share, or what you get in earnings for every dollar you invest.
- Forward P/E ratios, which use earnings estimates for one year into the future, can be more helpful in assessing the valuation of fast-growing healthcare stocks. Comparing P/E ratios with other stocks in the same industry will help you determine if the stock is relatively cheap or relatively expensive.
- Take growth prospects into consideration by checking out the stock’s price-to-earnings-to-growth (PEG) ratio, which incorporates projected earnings growth rates (typically over five years). Stocks with lower PEG ratios (especially when the ratios are less than 1) are more attractively valued than those with higher PEG ratios.
4. Dividends
- Some of the best healthcare stocks pay dividends -- a portion of earnings that the company returns to shareholders. Dividends can boost the overall return you receive from owning a stock.
- The dividend yield tells you how large a stock’s annual dividend payments are as a percentage of the current share price.
- Consider the stock’s payout ratio, which measures dividends as a percentage of earnings and indicates how much of the company’s cash is being used to cover the dividend. The lower the payout ratio, the greater the likelihood that the company will be able to keep paying dividends in the future.
What are the risks of investing in healthcare stocks?
Investing in any kind of stock comes with risks, including the possibility that competitors will develop more successful products and services. Healthcare stocks face these risks, as well as others that are more unique to the sector.
Healthcare is highly regulated. Drugmakers and medical device makers can fail to secure the necessary regulatory approvals to market new products. Regulatory changes can drastically alter a healthcare stock’s growth prospects. In the U.S., the Food and Drug Administration (FDA) oversees the regulation of drugs and medical devices. It’s smart to pay attention to any FDA action related to medical stocks you’re watching.
Many healthcare stocks also face significant litigation risk. For example, biopharmaceutical companies, medical device makers, and healthcare providers can be sued if patients think the companies’ products and services have caused them harm.
In addition, drugmakers and medical device makers must convince payers, including health insurers, PBMs, and government agencies, to buy their products. If companies aren’t successful in obtaining reimbursement approvals, their growth prospects can be reduced.
Many healthcare companies are also highly dependent on Medicare reimbursement levels. Recent changes to Medicare allow the program to negotiate prices for some high-cost drugs with pharmaceutical companies. Some drugmakers' revenues and profits could be negatively affected since Medicare pays less for certain drugs.
Benefits of investing in healthcare stocks
The key benefits of investing in healthcare stocks include:
- Many healthcare stocks have strong long-term growth prospects thanks to aging demographic trends.
- Healthcare stocks tend to perform well during economic downturns.
- These stocks provide diversification in one of the most important sectors.
- Many healthcare stocks offer attractive dividends.
Related investing topics
How to invest in healthcare stocks
Follow these steps to invest in healthcare stocks:
- Open your brokerage app: Log in to your brokerage account where you handle your investments.
- Search for the stock: Enter the ticker or company name into the search bar to bring up the stock's trading page.
- Decide how many shares to buy: Consider your investment goals and how much of your portfolio you want to allocate to this stock.
- Select order type: Choose between a market order to buy at the current price or a limit order to specify the maximum price you're willing to pay.
- Submit your order: Confirm the details and submit your buy order.
- Review your purchase: Check your portfolio to ensure your order was filled as expected and adjust your investment strategy accordingly.
Healthcare stocks should have healthy returns
Despite these risks, the overall outlook for healthcare stocks appears very good over the long term. Aging demographic trends around the world, combined with advances in technology, should open up tremendous opportunities for healthcare stocks -- and provide healthy returns for patient investors.