Phase 1
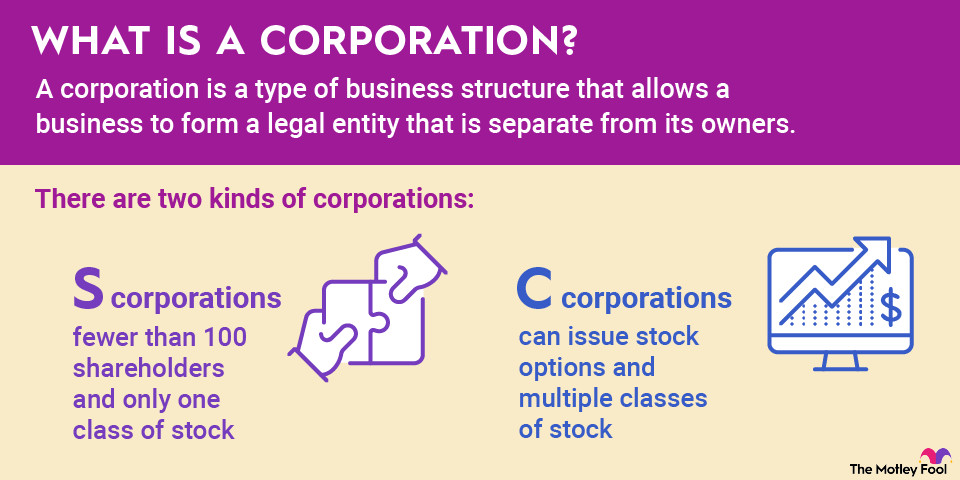
Phase 1 focuses on safety and is the most important phase to get off the ground. A small group of healthy volunteers (usually 20–100 people) receives the drug to determine safe dosage ranges and spot any side effects.
Phase 2
Phase 2 looks at effectiveness. Here, researchers test the drug on a larger group (100–300 people) who actually have the disease or condition. This phase helps confirm the treatment works and fine-tunes the dosage.
Phase 3
Phase 3 is where things scale up, and thousands of patients across multiple locations receive the drug. This phase compares the new treatment to existing options and monitors side effects across a larger population. This is often the final step before regulatory approval.
Phase 4
Phase 4 happens after the drug hits the market. Even after approval, ongoing studies track long-term effects, rare side effects, and overall performance in the general population.
Why clinical development phases matter
Pharmaceutical companies don’t necessarily like clinical phases, as, according to the Congressional Budget Office, it can cost up to $2 billion and take more than a decade from concept to approval. The main role of clinical phases is to protect patients, which also helps reduce risk and improve the chances of successful treatment. Without these stages, harmful or ineffective drugs could hit the market. Thalidomide, for example, was primarily used in Europe in the early 1960s, causing birth defects when taken during pregnancy. Cases like that helped lead to today’s stricter testing requirements.
Each phase helps to build trust from different parties in the underlying science behind the treatment or medicine. Regulators, doctors, and patients all rely on this framework to ensure treatments actually deliver on their promises. Pharmaceutical companies also depend on these results to gain patents, attract investors, and price their products appropriately. Remember: For pharmaceutical companies, government approval can be the difference between massive profits and billion-dollar write-downs. Mastering the clinical phase process is an absolute must.