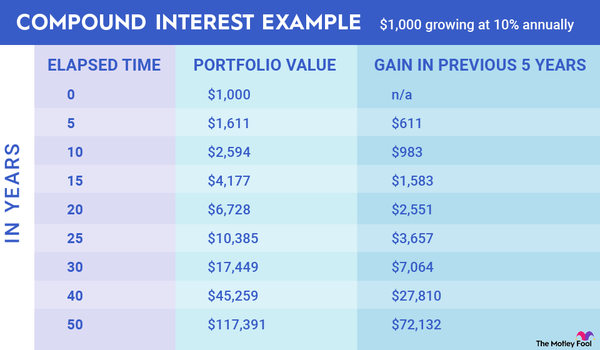
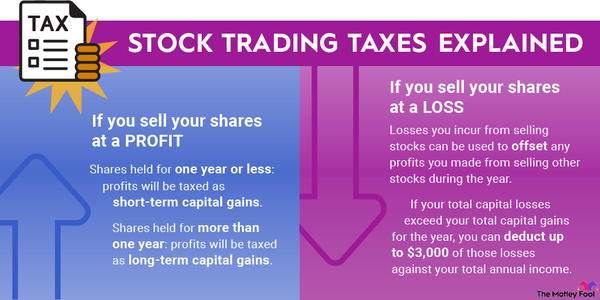
Eli Lilly (LLY -0.41%) is a leading pharmaceutical company. It has developed several blockbuster products, including Trulicity (type 2 diabetes), Taltz (plaque psoriasis), and Jardiance (blood sugar). These and other drugs generate billions in sales and profits for the company each year.
The drug company recently added a couple more blockbuster drugs to its portfolio by launching Mounjaro (diabetes) and Zepbound (obesity). They're already generating billions of dollars in revenue for the company.
The sales potential of these new drugs and the company's other therapies has many investors interested in the stock. Here's everything you need to know about how to invest in Eli Lilly.
Stock
How to buy
How to buy Eli Lilly stock
People interested in investing in Eli Lilly stock will need to take a few steps before becoming shareholders. This step-by-step guide will show you how to invest in stocks and add the pharmaceutical company to your portfolio.
- Open your brokerage app: Log in to your brokerage account where you handle your investments.
- Search for the stock: Enter the ticker or company name into the search bar to bring up the stock's trading page.
- Decide how many shares to buy: Consider your investment goals and how much of your portfolio you want to allocate to this stock.
- Select order type: Choose between a market order to buy at the current price or a limit order to specify the maximum price you're willing to pay.
- Submit your order: Confirm the details and submit your buy order.
- Review your purchase: Check your portfolio to ensure your order was filled as expected and adjust your investment strategy accordingly.
Should I invest?
Should I invest in Eli Lilly?
You need to decide whether investing in Eli Lilly is right for you. Here are some factors that could lead you to buy the pharmaceutical stock:
- You believe Zepbound has better sales potential than rival Novo Nordisk's (NVO 1.17%) Wegovy.
- You take one or more medications made by Eli Lilly.
- Adding Eli Lilly to your portfolio would help make it more diversified.
- You understand the risks of investing in pharmaceutical stocks, including the fact that they can lose money.
- You want to invest in a company that pays a growing dividend.
- You believe Eli Lilly can grow into its high valuation.
On the other hand, here are some reasons you might decide Eli Lilly isn't the right stock for you to buy:
- You're wary of pharmaceutical companies and prefer a more natural approach to health and wellness.
- You're unsure whether weight-loss drugs like Zepbound will live up to the hype.
- You're in or nearing retirement and need more dividend income than Eli Lilly can supply.
- You're concerned about Eli Lilly's high valuation (it had a forward price-to-earnings (P/E) ratio of more than 30 times in early 2025).
- You already own several pharmaceutical stocks.
- You're concerned about how U.S. Health and Human Services Secretary Robert F. Kennedy Jr. might affect the pharmaceutical industry.
Profitability
Is Eli Lilly profitable?
Digging into a company's profitability is essential because earnings tend to drive a stock's price over the long term. Given this dynamic, you'll want to see that the company is increasing its earnings or at least on the path toward profitability.
Eli Lilly is very profitable. The company reported $10.6 billion, or $11.71 per share, of net income in 2024 on $45 billion of revenue. Revenue was up 32% while earnings soared 102%.
The company expects to deliver another year of significant earnings growth in 2025. Its initial guidance has revenue growing to a range of $58 billion to $61 billion (32% growth at the midpoint), with earnings per share roughly doubling to between $22.05 and $23.55. New product sales are a big factor driving this view, including the continued growth of weight loss drug Zepbound and additional launches of Mounjaro outside of the U.S.
Dividends
Does Eli Lilly pay a dividend?
Eli Lilly has a long history of paying dividends. It has increased its payout annually for over a decade. It has been growing its payout at an accelerated pace in recent years, delivering its seventh straight year of hiking its dividend by 15% in late 2024:
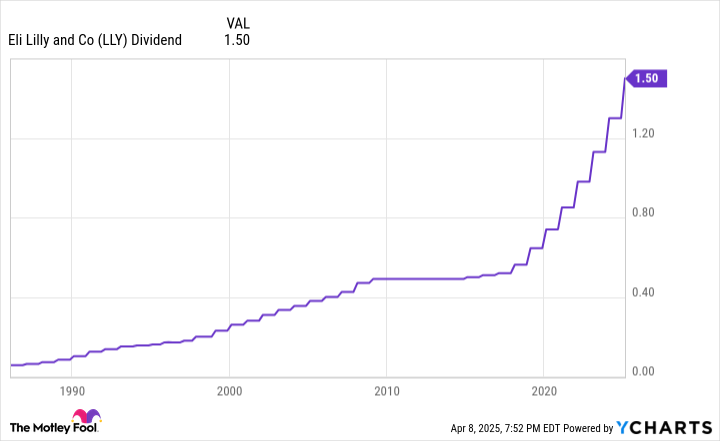
While the company pays a growing dividend, it offered a below-average dividend yield (0.8% in early 2025 compared to around 1.5% for the S&P 500).
ETF options
ETFs with exposure to Eli Lilly
Many investors prefer a passive approach to actively managing a portfolio of stocks. Exchange-traded funds (ETFs) make this easy. They allow you to passively invest in similar companies, market indexes, or other themes.
Exchange-Traded Fund (ETF)
According to ETF.com, 442 ETFs held 113.8 million shares of Eli Lilly in early 2025. The Vanguard Total Stock Market ETF (VTI 0.11%) was the largest holder, with 27 million shares. However, the broad market ETF had a rather small allocation to the pharmaceutical giant, at 1.4% of its holdings.
Investors seeking greater exposure to Eli Lilly have several ETF options, including those focused on top healthcare stocks:
- iShares U.S. Pharmaceutical ETF (IHE 0.98%): The fund tracks an index focused on U.S. pharmaceutical stocks. It held shares of 37 companies in early 2025, including Eli Lilly (second-largest at 23% of its net assets). The fund had a 0.39% ETF expense ratio.
- Health Care Select Sector SPDR Fund (XLV 1.35%): The fund tracks the healthcare sector of the S&P 500. The ETF had 60 holdings in early 2025, led by Eli Lilly at 11.6%, with a very low expense ratio of 0.08%.
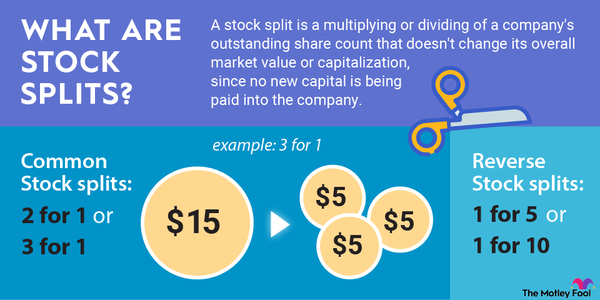
Stock splits
Will Eli Lilly stock split?
Eli Lilly didn't have an upcoming stock split as of early 2025. However, the pharmaceutical company has split its stock several times over the years. Here's a snapshot of Eli Lilly's stock split history:
| Split Year | Split Ratio |
| 1997 | 2-for-1 |
| 1995 | 2-for-1 |
| 1989 | 2-for-1 |
| 1986 | 2-for-1 |
| 1971 | 2-for-1 |
The company's last split came in 1997. Shares have gained significant value since then, trading at more than $700 in early 2025. Its high share price makes the stock less accessible to many investors, so Eli Lilly is an excellent candidate to complete a stock split in the future.
Related investing topics
The bottom line on Eli Lilly
Eli Lilly has grown into one of the world's largest pharmaceutical companies thanks to the blockbuster success of its drug development efforts. The company expects its revenue and profitability to continue rising, driven by new products like Zepbound and Mounjaro. That profit growth could send its stock even higher in the future, potentially making it a very rewarding long-term investment.
FAQ
Investing in Eli Lilly FAQ
Is Eli Lilly stock a good investment?
Eli Lilly stock has been a very good investment over the years. The company has delivered a total annual return of over 28% over the last decade, more than double the average S&P 500 return (over 11%). A big driver has been its success in developing blockbuster drugs.
The company's portfolio features several growth products and new drugs that should continue driving its revenue and profitability higher. Its profit growth should enable Eli Lilly to grow its dividend while potentially helping it to continue producing strong stock price appreciation.
However, the company does trade at a high valuation (more than 30 times forward earnings in early 2025). It can grow into that valuation as long as its new and growth products continue increasing sales and earnings at brisk rates.
How do I buy shares of Eli Lilly?
It's easy to buy shares of Eli Lilly. You need to open a brokerage account (if you don't have one already), figure out your budget, thoroughly research the company, and place an order when you're ready to buy shares. To place an order, you'd fill out the order page in your brokerage account, including:
- The number of shares you want to buy
- The correct stock ticker (LLY for Eli Lilly)
- The order type (limit order or market order)
Once you fill out the order page, click to submit your trade to buy shares of Eli Lilly.
Is Eli Lilly a good long-term stock?
Eli Lilly has historically been a good long-term stock investment. Over the last decade, the pharmaceutical stock has delivered an average annual return of 28%, more than double the return of the S&P 500 (around 11% annually).
While past performance is no guarantee that the company will deliver strong returns in the future, it has a lot going for it these days. Growth products like Jardiance, Taltz, and Trulicity are increasing sales rapidly. Meanwhile, new product launches like Mounjaro and Zepbound are off to a strong start and have become notable growth products.
Rising sales from these and other drugs should drive the company's profits higher, giving it more money to invest in R&D and return to shareholders through a growing dividend and share repurchases. Those factors have historically been key hallmarks of good long-term investments.
Who owns the most Eli Lilly stock?
The Lilly Endowment owned the most Eli Lilly stock in early 2025. It held more than 96 million shares (about 10.2% of its outstanding shares) worth roughly $87.1 billion. The Lilly Endowment is a private philanthropic foundation formed by J. K. Lilly, Sr., and his sons Eli and J. K., Jr., through gifts of stock in their pharmaceutical company, Eli Lilly.